Clinic Protocols for Receiving Reproductive Materials
Cryoport Systems has the proper solutions and procedures in place to ensure shipping compliance of your reproductive materials to clinics around the world. However, what happens once your materials reach their destination can seem highly varied from clinic to clinic. But no matter if the receiving clinic is located domestically or abroad, there are steps in place that clinics must take to properly receive your reproductive materials.
Cryoport Systems participated in the Cryogovernance V – Best Practices in Cryotransportation webinar in collaboration with The International IVF Initiative – i3 in October 2022. This webinar touched on important information of the standardized practices clinics must use to compliantly receive your reproductive materials. With information from Dr. Kristen Ivani, Seattle Reproductive Medicine’s Laboratory Director, we’re breaking down three important aspects of receiving cryogenic shipments that your receiving clinic should adhere to.
1. Examine Shipper for Damage
Once your shipment is received, clinical professionals need to first look for external damage before opening the shipper. Any external damage could indicate that the samples within the shipper have also been damaged. Dr. Ivani detailed three common indicators for clinic staff to look for that could indicate a damaged shipment.
- Opened shipper container – An opened shipper or container could mean that the package was tampered with prior to arrival.
- Damage to external box or shipper – Any dents, scratches, or cracks could have compromised the integrity of the shipment.
- Frost or sweating on the shipper – Moisture on the outside of the shipper could indicate damage to the shipper’s internal cooling agent.
The prospect of damage to shipments means that selecting a shipping partner who utilizes high-quality shippers is vital to maintaining your shipment’s integrity. Cryoport Systems’ Cryoport Express® shippers are specifically engineered with high-performance materials and advanced insulation to maintain the required temperature throughout the entire shipment. Our qualified shippers coupled with our supply chain management solutions can help ensure that your precious materials arrive safely and compliantly.
2. Review Attached Paperwork
All cryogenic shipments must be accompanied by paperwork. Dr. Ivani stated that those included forms will detail identifying information such as:
- Patient information like ID numbers and consent forms.
- Warming protocols for materials.
- Summary of records (SORs) and eligibility determination.
- Infectious disease screening results.
Clinic staff must review these forms prior to removing the samples to ensure that the identification materials match the sample. Your shipping provider should compile all necessary documentation to assist the clinic staff in moving forward with receiving the shipment.
Cryoport Systems offers end-to-end solutions for cryogenic logistics, including comprehensive documentation management services. Our team of experts can help clinics navigate the complex regulatory landscape and ensure that all necessary permits and certificates are in place for each shipment. This brings you added peace of mind that your samples will arrive at their destination with all the proper documentation.
3. Prepare Proper Storage Location
Once external inspections of the shipment and the verification of documents have been completed, the clinic staff must prepare the proper storage location for your materials. Your precious materials must be stored in a secure and controlled environment to maintain their viability and integrity.
The process of removing your samples from the shipper and moving them to their storage location must be completed quickly and compliantly. The storage location should be equipped with appropriate cryogenic storage systems such as liquid nitrogen freezers or tanks. Once the shipment has been removed from the shipper and placed in storage, it’s important that your clinic has monitoring systems that are regularly checked and maintained to ensure the integrity of your materials.
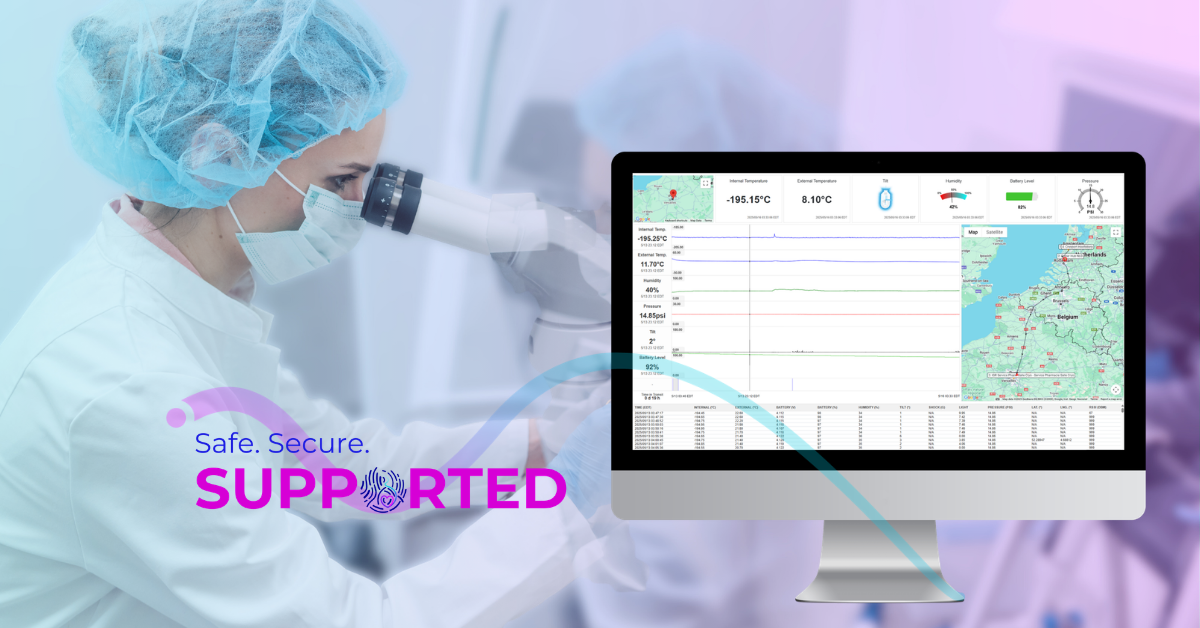
Throughout the shipment process, your precious materials will be monitored by the Cryoport Solution when shipping with Cryoport Systems. Our intelligent solutions monitor the integrity of your shipment for information on its location, internal temperature, orientation, and more. With this specific information, the receiving clinic will have record of any risk that could have befallen your materials even before they’re moved to storage.
Receiving cryogenic shipments of reproductive materials is a critical task that requires strict adherence to proper protocols. By working with trusted shipping partners like Cryoport Systems, clinics can access the expertise and resources they need to manage cryogenic logistics with confidence. We encourage you to speak with your current and potential receiving clinic if you have specific questions about their procedures.