Protecting Reproductive Materials from Origin to Destination
Reproductive materials differ from everyday commodities that move through a logistics network. Their viability depends on narrow, well‑understood thermal conditions, and their value extends far beyond a clinical procedure. When clinics ship oocytes, sperm, embryos, and other sensitive materials, they trust that the system responsible for protecting them will behave consistently under real‑world conditions. That trust is not earned through equipment alone. It comes from evidence that the system performs predictably across a wide range of scenarios every time it is used.
Validated shipping systems provide that evidence. They show how a shipper behaves across temperature exposures, transport durations, carrier handoffs, and environmental variations. Most importantly, they allow clinics to rely on performance rather than assumptions. In a field where a few degrees or a few hours can influence outcomes, that reliability becomes a critical part of patient care.
Cryoport Systems’ shipping systems are built specifically for biological materials and validated to ensure the stability and predictability that clinics depend on. They work in concert with ISO 21973‑certified processes and continuous monitoring to maintain material integrity from preparation through final delivery. Together, these elements form a controlled pathway through transit, one that provides clinics, embryologists, and intended parents with confidence that the materials were protected at every step.
Why Reproductive Materials Need Predictable, Proven Protection
The cryogenic environment is often discussed as though it guarantees safety, but stable temperature alone is not enough. Liquid nitrogen vapor shippers perform well when they are engineered correctly, maintained consistently, and prepared through controlled procedures. When any part of that chain varies (equipment, conditioning time, loading technique, or handling, for example), the performance curve can shift.
Reproductive materials are especially sensitive to variability. Clinics see the impact of this most clearly when they attempt to reconstruct what happened during a shipment that did not arrive as planned. The equipment may appear intact, yet the data reveals dips in thermal behavior or timing irregularities that are difficult to interpret without validated performance parameters.
Because reproductive logistics often involves multiple organizations, each with its own tools and documentation practices, clinics must be able to rely on the shipping system itself (not an assortment of logs) to preserve viability. A validated system removes ambiguity by demonstrating in advance how that system behaves under defined conditions. It provides a factual reference point for interpreting monitoring data and understanding performance margins. For clinics and intended parents navigating emotionally and clinically significant decisions, that clarity matters.
Closing Gaps with Requalification and Validation
One of the most persistent sources of risk in reproductive logistics is the assumption that a cryogenic shipper will perform the same way every time it is used. In reality, performance can drift when equipment is reused without a controlled process to verify that it still meets its original specifications. Materials as sensitive as embryos and oocytes cannot be protected by a shipping system whose behavior is taken on trust. They require equipment that has been proven, cycle after cycle, to perform within predictable boundaries.
Cryoport Systems addresses this by requalifying every shipping system before each use. This is not a surface‑level inspection or a quick conditioning cycle. It is a structured process designed to confirm that the shipper is functioning at the level originally demonstrated during validation. Every unit goes through a controlled thermal preparation sequence, handled under the same conditions and timing that were used to establish its performance criteria. This approach eliminates guesswork about how long the shipper will maintain its environment and how it will behave once it enters transit. Clinics receive equipment that has been brought back to a known state, not equipment whose performance depends on prior handling or residual conditions.
A critical part of this process is Veri‑Clean®, Cryoport Systems’ controlled decontamination method. After every use, each shipper undergoes a validated cleaning procedure designed specifically for cryogenic transport. This removes contaminants that can accumulate through handling, exposure, and carrier processing environments. It also eliminates the variability introduced when different teams rely on their own cleaning practices. Veri‑Clean standardizes this step across the network, ensuring that every unit begins its next journey in a known, controlled state. For reproductive materials, where even minor contamination can have significant consequences, this level of attention is essential.
Most shippers available in the market are neither requalified nor decontaminated through a validated process before each use. Some are cleaned based on visual inspection, others conditioned informally, and many put back into circulation without a documented verification step. That variability introduces uncertainty into the Chain of Compliance® and makes it more difficult for clinics to interpret the performance of the equipment when reviewing monitoring data. By contrast, Cryoport Systems’ approach removes that uncertainty. A clinic receiving a Cryoport Systems shipping system knows that the equipment has been fully decontaminated, prepared under controlled conditions, and requalified to operate within its validated parameters. Not only that, but full equipment and performance history of all shipping systems and shipments are documented and controlled within our Cryoportal® logistics management system for an audit-ready historical record of every shipment and every shipping system.
Requalification and validation, combined with Veri‑Clean and ISO 21973‑certified processes, create a closed loop of control around each shipment. It is an approach built specifically for reproductive medicine, where predictability and protection must extend through the entire journey, not just at the point of departure. Cryoport Systems uses this approach for every shipper, every time, because the materials entrusted to these systems leave no margin for uncertainty.
Continuous Monitoring as an Added Safeguard with Expert Oversight and Intervention When Needed
Monitoring is important, but its role is often misunderstood. It cannot correct weaknesses in equipment design or preparation, and it cannot compensate for inconsistent handling. Its primary purpose is to verify that a validated system is performing within its expected parameters. When monitoring is paired with a stable, predictable shipping platform, it becomes a valuable safety net.
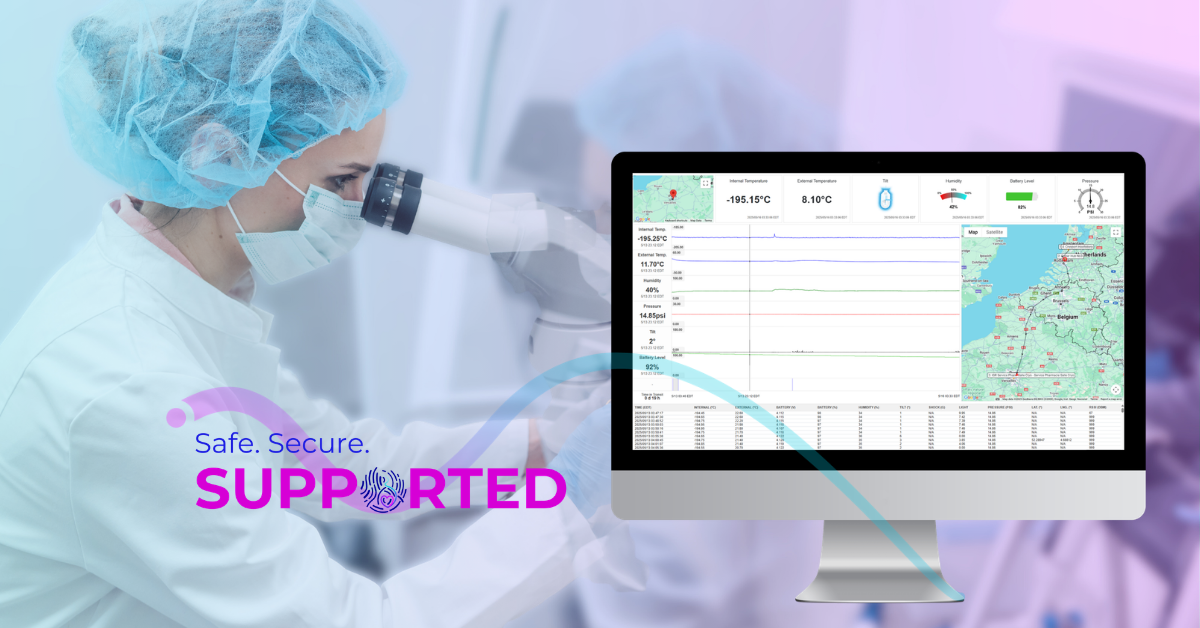
Cryoport Systems uses continuous monitoring to capture environmental and performance data (temperature, humidity, tilt, shock, and other critical parameters) throughout transit. This information is not limited to a final report; it is reviewed during the journey and used to inform decisions when shipping conditions shift. If a route is delayed, weather changes, or carrier performance indicates increased risk, the monitoring data helps determine the best course of action. It turns oversight into a proactive function rather than a reactive one.
Even validated systems encounter challenges in the real world. Flights are delayed, weather shifts unexpectedly, and packages can be held longer than planned in clinical facilities. Cryoport Systems provides dedicated oversight to manage these events.
When monitoring data indicates a change in risk, trained specialists review the situation and take appropriate action. This may involve adjusting the route, coordinating with carriers, or escalating through internal procedures to protect the material. The key is that interventions follow defined protocols, informed by both monitoring data and knowledge of the system’s performance limits.
This layer of oversight complements the validated platform. The system is designed to perform reliably, and the monitoring ensures that performance aligns with expectations. When external factors threaten that stability, expert intervention provides an additional safeguard.
For clinics, a validated shipping system supported by continuous monitoring and expert oversight simplifies both clinical and operational work. It reduces the uncertainty associated with transit, allows teams to plan with confidence, and minimizes the likelihood of outcomes influenced by preventable issues.
Reproductive materials deserve a shipping system engineered to protect them, supported by processes proven through validation, and monitored by experts who understand what is at stake. Cryoport Systems’ validated shipping systems provide that foundation. They offer the stability, predictability, and oversight needed to maintain the integrity of sensitive materials from origin to destination.
As documentation expectations rise and clinical standards evolve, programs that rely on validated, evidence‑based shipping systems will be better positioned to maintain reliability and support the outcomes that matter most to patients and providers. Cryoport Systems remains committed to delivering that level of protection through every shipment, every route, and every interaction.