How Shipment Visibility Builds Trust Across Clinic Networks
For clinics supporting reproductive medicine, trust is rarely lost through failure alone. More often, it erodes through uncertainty. Clinics may have strong internal controls and experienced teams, yet still find themselves unable to answer basic questions once a shipment of reproductive materials (such as embryos, oocytes (eggs), or sperm) leaves the building. When visibility diminishes outside the lab, confidence soon follows.
For single-site clinics, this gap can be frustrating. For multi-clinic networks, it becomes structural. Shipments may be moving simultaneously across regions, while clinic organizations must be able to demonstrate that transport is governed with the same rigor applied to laboratory processes. When the shipment status and condition history are fragmented across tools or vendors, clinics are often left reconciling what may have happened after the fact, rather than operating from a defensible, shared source of truth.
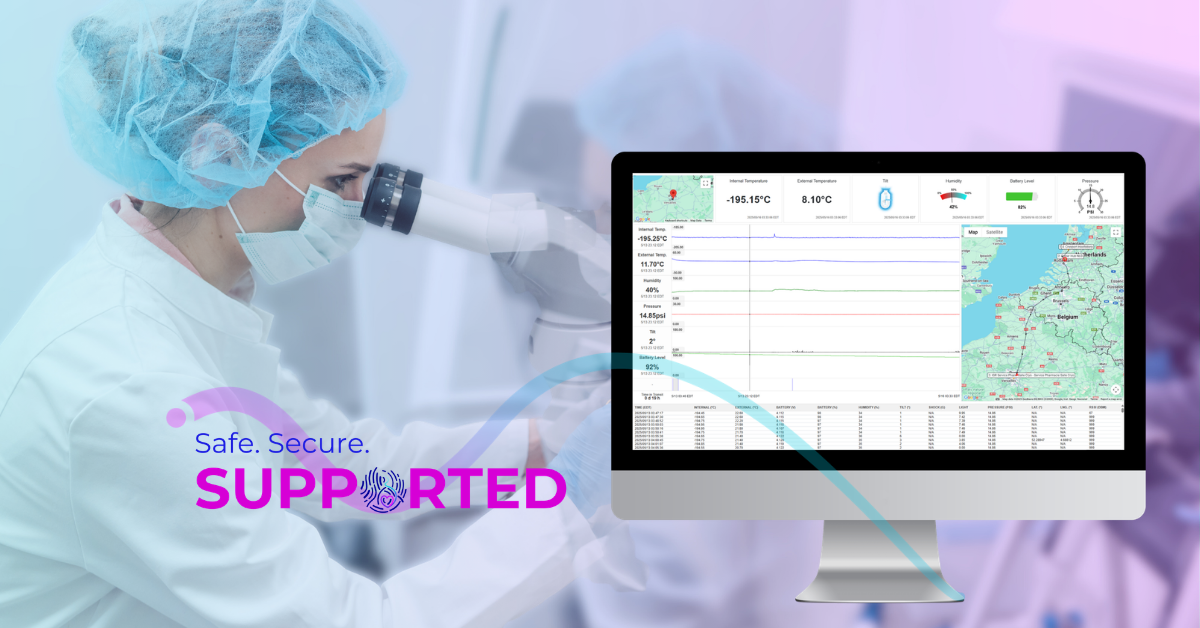
Visibility closes that gap. When the clinic can clearly see where materials are, how they are being handled, and under what conditions (temperature, humidity, tilt, shock, etc.), transparency becomes the mechanism that sustains trust across networks. The result is not simply smoother logistics, but greater confidence in compliance and accountability, supporting more credible and consistent communications with patients at moments when certainty matters most.
Where Clinic Networks Feel the Strain
As clinic networks expand, shipment visibility tends to fragment. Each site may follow sound internal practices, but transport often sits at the intersection of various systems, vendors, and site-specific workflows. What works well for a single location becomes harder to sustain when dozens of shipments are moving simultaneously across different geographies and regulatory environments.
For many networks, visibility relies on a patchwork of status updates and standalone emails. The information exists, but not in a standardized or centralized format. While clinics may be able to reconstruct a shipment’s journey later (through a tangle of different emails, portals, and tracking numbers), it can be difficult to gain a clear picture while materials are in motion. All of this creates operational drag, slowing teams down as they spend time chasing answer, and confidence becomes tied to individual follow-ups rather than a consistent process.
This is where standardized vendor relationships and shipment visibility becomes essential as shared infrastructure across the network. When every clinic operates from the same system and standard, audit-ready compliance records that track every shipment (including shipment details, condition monitoring, and compliance records) allow transparency to scale together with growth. Clinics gain predictability and transportation of reproductive materials becomes a governed extension of the clinical environment (rather than an external blind spot).
Operationalizing Visibility
Visibility at this level does not happen by accident. It requires logistics partners that are built to support reproductive medicine, with systems that have been intentionally engineered to capture and preserve shipment data as part of routine operations. For clinic networks, this level of visibility and accountability matters. While regulatory standards for reproductive medicine have lagged behind those of other forms of human medicine (like cell and gene therapies), they are starting to catch up. Clinics can plan now for future-proofed transportation, proactively turning shipping into a process that adheres to the highest levels of compliance and documentation.
Cryoport Systems supports this through clinic-facing visibility tools that are integrated by design. Continuous condition monitoring tracks shipment environments and conditions throughout transit, while the Cryoportal® logistics management platform provides a centralized view of active and historical shipments across sites. Together with the Chain of Compliance®, each shipment generates a complete and audit-ready record of custody, condition, and handling from origin to destination. Visibility is built into the process itself rather than relying on manual follow-ups or individual familiarity with a given shipment.
For clinic networks, this consistency is critical. Every clinic operates from the same standards and documentation framework, and shipment data is accessible when decisions need to be made rather than reconstructed later for reporting or audit purposes. This creates a solid foundation where transport aligns with laboratory governance, where compliance expectations are met as a matter of course.
This level of transparency also changes how clinics communicate. When shipment visibility is clear and shared, updates are factual and data-driven with the confidence that comes from evidence rather than assumption. Teams know with absolute confidence that the materials were maintained within temperature specifications from origin to destination with no deviations in conditions or handling. That level of clarity supports clinical teams and patient-facing staff alike, enabling standardized workflows and credible conversations at moments where precision matters.
Translating Infrastructure into Outcomes
From an operational standpoint, this transparency reduces variability across the network. Shipment processes do not rely on local workaround knowledge or individual judgement, instead following a defined and repeatable structure that applies equally to every shipment across every site. This consistency makes it possible to accurately assess performance and identify trends, addressing any issues systemically rather than anecdotally.
From a compliance perspective, shipment visibility simplifies what is often one of the most resource-intensive responsibilities… documentation. Audit-ready records are generated as shipments move, not assembled retroactively under time pressure. Condition data, custody records, and shipment parameters are captured automatically and preserved in a single system. And when questions arise, internally or externally, clinics can respond with documented evidence rather than reconstructed timelines that were pieced together after the fact.
As regulatory expectations evolve and accreditation bodies place greater emphasis on transport controls, clinics that already operate under standardized governance (such as ISO 21973-certified processes) are better positioned to adapt without disruption. Visibility shifts compliance from a reactive effort to a baseline capability when clinics have the right vendor partnerships in place.
Transport then becomes what it should be, a controlled extension of clinical practices. When shipment visibility is continuous and documented, clinics gain confidence not only in where materials are going but also in their ability to demonstrate how those materials were protected every step of the way.
Building Trust in the Patient Experience
This confidence does not stay confined within logistics or compliance teams. It shapes how clinics engage with patients during some of the most emotionally charged moments of care. When shipment visibility is well governed, patient communications are no longer built on reassurance alone, they are built on facts.
Patient-facing teams do not need to speculate or soften uncertainty. They can explain where materials are and how they were protected during transit. That clarity matters. Intended parents may never see the underlying platforms or condition data, but they feel the difference when messages are consistent and grounded in documented reality.
For clinic networks, this transparency supports a more uniform patient experience across locations. Care does not feel dependent on which clinic initiates a shipment or which staff member happens to be on call. Instead, communication standards are reinforced by shared visibility and documented processes.
In reproductive medicine, where materials are irreplaceable and expectations are (rightly) high, shipment visibility is a core component of quality. By implementing transparent, standardized shipping practices, clinics strengthen accountability and reduce uncertainty, allowing them to build trust at scale. Trust within their teams, with regulators, and – most importantly – with the patients they serve.