Extending Clinical Accountability Beyond the Lab
In reproductive medicine, excellence inside the lab is expected. Where clinics are increasingly judged is the space between buildings… what happens after a specimen leaves controlled hands and whether the clinic can demonstrate that the same discipline traveled with it. When transport is treated as a wholly external utility, the evidentiary trail fractures and avoidable risk can turn into organizational drag. A clinic that cannot show custody, identity, and environmental history with confidence is left reconstructing what they think happened with a shipment, rather than operating from demonstrated absolutes.
If accountability is the norm inside the laboratory, it must travel with the specimen. Proactive governance makes that expectation explicit across storage, movement, and long-term stewardship. And with the Chain of Compliance®, Cryoport Systems turns that philosophy into an operational reality for transport, enabling clinics to provide governed shipments that generate complete, audit-ready proof by default.
The Hidden Consequences of Unseen Errors
What puts clinics at risk during transport isn’t only what might go wrong in motion, it’s also what can’t be demonstrated afterward. When chain of custody, chain of identity, and chain of condition aren’t captured as one continuous record, a solvable deviation becomes a credibility problem that invites scrutiny and erodes trust. Recent legal perspectives in the field have been clear that stronger controls are no longer optional, as carriers and insurers increasingly expect electronic witnessing, track-and-trace across the entire process, continuous monitoring of critical materials, and even relationships with specialized logistics partners who operate to those standards.
This shift reflects a broader move from reactive management to cryogovernance. Clinics are increasingly unifying their policies across the end-to-end workflow, and, as a result, it’s important that evidence and data stay intact from bench to biorepository to courier and back again. This isn’t to create more paperwork, but to create a single source of truth that stands up when a patient asks for the record or an auditor requests proof months later. For clinics, treating transport as an extension of the controlled clinical space adds certainty and removes the guesswork of verbal handoffs and scattered, disconnected documentation. It allows teams to prove how materials were handled at every step and within what conditions.
From Risk Management to Governance
Many clinics still run transport they way they ran storage a decade ago. Diligent people and respectable processes, but documentation scattered across systems that were never designed to be read as a single, comprehensive narrative. That was workable when volumes were lower and workflows were simpler, but it’s not sustainable in today’s environment. Clinics now navigate multi-site networks, with increasing patient populations and a growing need to transport reproductive materials, alongside increasing scrutiny as regulations try to catch up and keep pace with industry developments.
Governance, applied to transport, is simple to state and hard to fake. The story of material handling lives inside the same account that follows the journey. The safeguards that protect chain of custody and chain of identity are recorded in that account rather than parked in a parallel spreadsheet or email chain. The conditions experienced during transit are captured without gaps and tied to the same shipment identity, so clinics don’t guess at what happened while materials were out of sight.
Chain of Compliance® Makes Transport Clinically Defensible
Chain of Compliance is Cryoport Systems’ proprietary framework for transport governance. This purpose-built structure keeps the evidentiary integrity of a shipment intact, unifying what most clinics struggle to connect: chain of custody, chain of identity, and chain of condition.
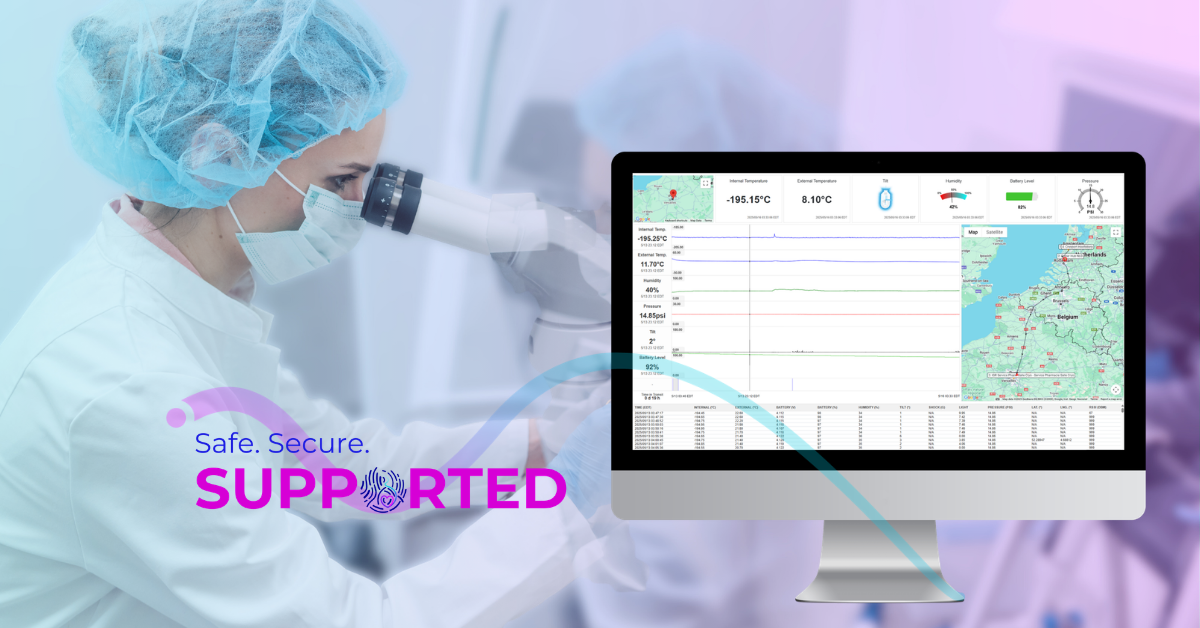
At a practical level, Chain of Compliance fills the gaps for documentation that clinics cannot replicate on their own across third-party movement. It creates a single shipment history that begins before loading and persists after delivery. Equipment histories as well as performance and deviation records belong to the shipment’s account. It maintains continuous evidentiary controls while the shipment is in transit, correlating in-field events with equipment performance and preserving those correlations as part of the record. It makes the record durable, with calibration, maintenance, requalification, and sanitization steps not only performed but also documented in a way that can be retrieved and read as a single narrative after the transfer. It ensures that continuous monitoring throughout transit tracks critical parameters like temperature, tilt, shock, and humidity for documented assurance that conditions remained consistent at every step from origin to destination.
In the past, clinics leaned on internal SOPs and the occasional data logger. But today, shipment proof has to cross boundaries without breaking, and do so every time, for every shipment. A clinic can control how materials are prepared and handed off, but it cannot, by itself, generate an auditable history of the equipment, performance, deviations, and custody events that occur in transit. Chain of Compliance exists to solve exactly that gap, and it’s why clinics that adopt it stop reconstructing stories and instead simply retrieve them.
What it Looks Like When Transport is Governed
Picture, if you will, a weekday transfer from storage to clinic. Under traditional handling, the team prepares the materials, confirms identity, hands off to a courier, and saves a tracking number. Condition data sits on a device that someone will download later. If weather forces a route change, people make calls, send emails, and keep notes. If a patient asks next week what happened during a particular interval, the clinic assembles the story from the various emails, local files, and memories.
Under Chain of Compliance, the same transfer reads differently. The shipment record begins before loading. As custody changes, that change becomes part of the same history, not a line in an inbox. The conditions materials experienced on the journey are recorded in connection with the shipment identity, not as a standalone device report. If timelines shift and actions are taken to protect the shipment, those actions are documented within the same record. At receipt, the clinic doesn’t rebuild a timeline; it simply receives one. Months later, the same account answers an audit with the same clarity it provided the patient.
When the story is built into the process, routine inquiries don’t spiral. The clinic isn’t tasked with investigating and rebuilding an entire shipment history, but instead can simply pull up the record. Not only does this protect clinics and patients alike, but it also enables scale. Multi-site programs carry variability by definition, but a uniform evidentiary standard for transport prevents that variability from compounding.
Transport as Part of the Quality Approach
Integrating transport into clinical governance doesn’t require upheaval. It requires aligning the evidence with the process that produces it. Start by defining, in plain terms, what “defensible” transportation of reproductive materials means for your program. If that definition includes a single account that contains custody events, identity verification, and the environmental story from preparation through receipt, you have described the standard that Chain of Compliance is built to deliver, and it’s time to partner with Cryoport Systems.
In the end, governed transport isn’t about adding another layer of compliance to an already demanding workflow. Rather, it’s about aligning the movement of patient materials with the same standard of accountability that defines modern IVF practice. When clinics adopt a framework like Chain of Compliance, they stop relying on hope and instead rely on evidence that stands up to the weight of clinical operations and legal expectations. That shift allows leaders to run programs that can grow without compounding risk while operating with the confidence that nothing in the lifecycle of a specimen is left to interpretation. That is the future cryogovernance calls for, and the standard the best clinics will hold.