Reproductive Medicine Archives

03/19/2026
Protecting Reproductive Materials from Origin to Destination
Reproductive materials differ from everyday commodities that move through a logistics network. Their viability depends on narrow, well‑understood thermal conditions, and their value extends far beyond a clinical procedure. When clinics ship oocytes, sperm, embryos, and other sensitive materials, they trust that the system responsible for protecting them will behave consistently under real‑world conditions. That trust is not earned through equipment alone. It comes from evidence that the system performs predictably across a wide range of scenarios every time it is used.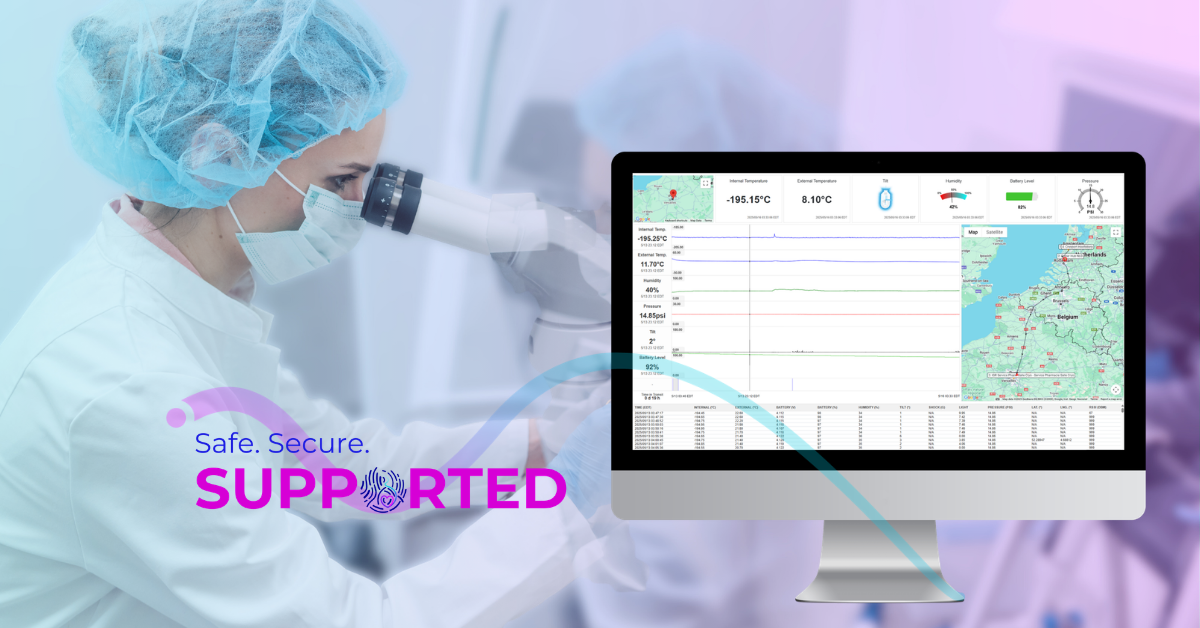
02/24/2026
Extending Clinical Accountability Beyond the Lab
In reproductive medicine, excellence inside the lab is expected. Where clinics are increasingly judged is the space between buildings… what happens after a specimen leaves controlled hands and whether the clinic can demonstrate that the same discipline traveled with it. When transport is treated as a wholly external utility, the evidentiary trail fractures and avoidable risk can turn into organizational drag.
02/03/2026
Why ISO 21973 Matters for Reproductive Transport: Setting the Standard for Safety
When reproductive materials leave the controlled environment of a clinic, the conditions they encounter can be unpredictable. A shipment might sit on a tarmac in extreme heat, pass through multiple couriers, or face delays at customs. Each of these moments introduces risk. And for patients, the stakes couldn’t be higher. That’s why standards like ISO 21973 matter.
11/18/2025
Risk in Practice: The Embryologist’s Frontline Safeguards
When it comes to fertility treatments and IVF, embryologists are often seen as the final line of defense, the protectors of patient specimens who ensure that all patient materials are handled with precision. But as Amanjot Grewal (Scientific Director, Pinnacle Egg Bank) emphasized during the Cryogovernance® VIII webinar, safeguarding reproductive materials is not only about technical skill. It’s about systems. It’s about governance. And it’s about recognizing that even the most diligent embryologist cannot mitigate risk alone.
11/05/2025
Managing Risk at Scale with Lessons from Network Laboratories
For modern IVF clinic networks, growth is both a measure of success and a new source of complexity. Expanding to multiple clinics and laboratories enables providers to reach more patients and offer consistency in care, but as these networks scale, so do the challenges of maintaining standardization and safety with full traceability across every site.
10/21/2025
Redefining Risk in IVF and Why Cryogovernance® Matters
Reproductive medicine is evolving faster than ever. The demand for in vitro fertilization (IVF) has surged across continents, driven by expanded access and new technologies as well as greater global mobility among patients. But that same growth has amplified the challenges (and risks) facing IVF programs worldwide. The truth is that IVF success today depends as much on safe, reliable cryostorage and transport as it does on laboratory technique. Yet as laboratories scale operations and manage increasingly complex storage networks, the systems in place to protect those sensitive materials haven’t always kept pace.Categories
- All (49)
- Ask the CryoStork® (10)
- Fertility Clinics and Care Providers (6)
- Patient Advocacy and Awareness (24)
- Success Stories (4)
- What to Expect When Shipping (31)
